cho 10,2 gam al2 o3 tác dụng hết với hcl viết phương trình phản ứng
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


Ta có: \(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
\(n_{Al_2O_3}=\dfrac{10,2}{102}=0,1\left(mol\right)\)
PT: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O\)
a, Theo PT: \(n_{HCl}=3n_{Al}+6n_{Al_2O_3}=1,2\left(mol\right)\)
\(\Rightarrow m_{HCl}=1,2.36,5=43,8\left(g\right)\)
b, Theo PT: \(n_{H_2}=\dfrac{3}{2}n_{Al}=0,3\left(mol\right)\)
\(\Rightarrow V_{H_2}=0,3.22,4=6,72\left(l\right)\)
\(n_{AlCl_3}=n_{Al}+2n_{Al_2O_3}=0,4\left(mol\right)\)
\(\Rightarrow m_{AlCl_3}=0,4.133,5=53,4\left(g\right)\)

\(n_{Al_2O_3}=\dfrac{10,2}{102}=0,1\left(mol\right)\)
PTHH: Al2O3 + 3H2SO4 → Al2(SO4)3 + 3H2
Mol: 0,1 0,1
\(m_{Al_2\left(SO_4\right)_3}=0,1.342=34,2\left(g\right)\)

1.
\(m_{HCl}=\dfrac{10,95.75}{100}=8,2125\left(g\right)\)
\(\Rightarrow n_{HCl}=\dfrac{8,2125}{35,5}=0,225\left(mol\right)\)
\(Fe_2O_3+6HCl\rightarrow2FeCl_3+3H_2O\)
\(\Rightarrow n_{Fe_2O_3}=\dfrac{1}{6}n_{HCl}=0,0375\left(mol\right)\)
\(n_{FeCl_3}=\dfrac{1}{3}n_{HCl}=0,075\left(mol\right)\)
\(\Rightarrow C\%=\dfrac{0,075.162,5}{0,0375.160+75}.100\%=15,05\%\)

a. \(n_{Al_2O_3}=\dfrac{10,2}{102}=0,1\left(mol\right)\)
PTHH : Al2O3 + 6HCl -> 2AlCl3 + 3H2O
0,1 0,6 0,2 ( mol )
\(m_{HCl}=0,6.36,5=21,9\left(g\right)\)
\(m_{AlCl_3}=0,2.133,5=26,7\left(g\right)\)
b.
PTHH : 3O2 + 4Al -> 2Al2O3
0,15 0,1 ( mol)
\(V_{O_2}=0,15.22,4=3,36\left(l\right)\)
a. \(n_{Al_2O_3}=\dfrac{10,2}{102}=0,1\left(mol\right)\)
PTHH : Al2O3 + 3HCl -> 2AlCl3 + 3H2O
0,1 0,3 0,2 ( mol )
\(m_{HCl}=0,3.36,5=10,95\left(g\right)\)
\(m_{AlCl_3}=0,2.133,5=26,7\left(g\right)\)
b.
PTHH : 3O2 + 4Al -> 2Al2O3
0,15 0,1 ( mol)
\(V_{O_2}=0,15.22,4=3,36\left(l\right)\)

n H2SO4=\(\dfrac{10\%.490}{2+32+16.4}=0,5mol\)
n Al2O3 =\(\dfrac{10,2}{27.2+16.3}=0,1mol\)
\(Al_2O_3+3H_2SO_4->Al_2\left(SO_4\right)_3+3H_2O\)
bđ 0,1............0,5
pư 0,1............0,3..................0,1
spu 0 ................0,2................0,1
=> sau pư gồm H2SO4 dư , Al2(S04)3 và H2O
m H2SO4 dư = \(0,2.\left(2+32+16.3\right)=19,6g\)
m Al2(SO4)3 = \(0,1\left(27.2+32.3+16.4.3\right)=34,2g\)
m dd = \(490+10,2=500,2g\)
% Al2(SO4)3 = \(\dfrac{34,2}{500,2}.100\sim6,84\%\)
% H2SO4 dư = \(\dfrac{19,6}{500,2}.100\sim3,92\%\)

Phản ứng xảy ra:
\(2Al+6HCl\rightarrow2Alcl_3+3H_2\)
\(Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O\)
Ta có:
\(n_{H_2}=\frac{3,36}{22,4}=0,15mol\)
\(\rightarrow n_{Al}=\frac{2}{3}n_{H_2}=0,1mol\)
\(\rightarrow m_{Al}=0,1.27=2,7gam\rightarrow m_{Al_2O_2}=10,2gam\)
\(\rightarrow\%m_{Al}=\frac{2,7}{12,9}=20,93\%\rightarrow\%m_{Al_2O_2}=79,07\%\)
\(n_{Al_2O_3}=\frac{10,2}{27.2+16.3}=0,1mol\)
\(\rightarrow n_{HCl}=3n_{Al}+6n_{Al_2O_3}=0,1.3+0,1.6=0,9mol\)
\(\rightarrow m_{HCl}=0,9.36,5=32,85gam\)
\(\rightarrow m_{ddHCl}=\frac{32,85}{3,65\%}=900gam\)

\(n_{Al2O3}=\dfrac{10,2}{102}=0,1\left(mol\right)\)
Pt : \(Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O|\)
1 6 2 3
0,1 0,6 0,1
a) \(n_{HCl}=\dfrac{0,1.6}{1}=0,6\left(mol\right)\)
\(m_{HCl}=0,6.36,5=21,9\left(g\right)\)
\(m_{ddHCl}=\dfrac{21,9.100}{10}=219\left(g\right)\)
b) \(n_{AlCl3}=\dfrac{0,6.2}{6}=0,2\left(mol\right)\)
⇒ \(m_{AlCl3}=0,2.133,5=26,7\left(g\right)\)
c) \(m_{ddspu}=10,2+219=229,2\left(g\right)\)
\(C_{AlCl3}=\dfrac{26,7.100}{229,2}=11,65\)0/0
Chúc bạn học tốt
nAl2O3=10.2:102=0.1(mol)
PTHH:Al2O3+6HCl->2AlCl3+3H2O
theo pthh:nHCl:nAl2O3=6->nHCl=6*0.1=0.6(mol)
mHCl=0.6*36.5=21.9(g)
mdd HCl=21.9*100:14.6=150(g)
theo pthh:nAlCl3:nAl2O3=2->nAlCl3=0.1*2=0.2(mol)
mAlCl3=0.2*133.5=26.7(g)
mdd sau phản ứng:10.2+150=160.2
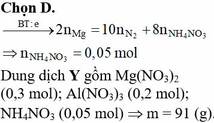
Pt: Al2O3 + 6HCl -> 2AlCl3 + 3H2O