Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, \(3Fe+2O_2\underrightarrow{t^o}Fe_3O_4\)
\(2Cu+O_2\underrightarrow{t^o}2CuO\)
\(4Al+3O_2\underrightarrow{t^o}2Al_2O_3\)
\(2Zn+O_2\underrightarrow{t^o}2ZnO\)
\(2Mg+O_2\underrightarrow{t^o}2MgO\)
\(2Ca+O_2\underrightarrow{t^o}2CaO\)
\(4Na+O_2\underrightarrow{t^o}2Na_2O\)
\(4K+O_2\underrightarrow{t^o}2K_2O\)
b, \(S+O_2\underrightarrow{t^o}SO_2\)
\(C+O_2\underrightarrow{t^o}CO_2\)
\(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
\(2H_2+O_2\underrightarrow{t^o}2H_2O\)
c, \(CH_4+2O_2\underrightarrow{t^o}CO_2+H_2O\)
\(C_2H_4+3O_2\underrightarrow{t^o}2CO_2+2H_2O\)
\(C_2H_6+\dfrac{5}{2}O_2\underrightarrow{t^o}2CO_2+3H_2O\)
\(C_3H_8+5O_2\underrightarrow{t^o}3CO_2+4H_2O\)

a) C+O2---->CO2
4P+5O2--->2P2O5
3Fe+2O2--->Fe3O4
2Mg+O2--->2MgO
4Al+3O2--->2Al2O3
2H2+O2--->2H2O
b) CH4+2O2--->CO2+2H2O
C4H10+5/2O2--->4CO2+5H2O
C2H6O+3O2---->2CO2+3H2O

\(C+O_2\xrightarrow[]{t^o}CO_2\)
\(S+O_2\xrightarrow[]{t^o}SO_2\)
\(4P+5O_2\xrightarrow[]{t^o}2P_2O_5\)
\(H_2+\dfrac{1}{2}O_2\xrightarrow[]{t^o}H_2O\)
\(4Na+O_2\rightarrow2Na_2O\)
\(Mg+\dfrac{1}{2}O_2\xrightarrow[]{t^o}MgO\)
\(4Al+3O_2\xrightarrow[]{t^o}2Al_2O_3\)
\(3Fe+2O_2\xrightarrow[]{t^o}Fe_3O_4\)
\(Cu+\dfrac{1}{2}O_2\xrightarrow[]{t^o}CuO\)
\(CH_4+2O_2\xrightarrow[]{t^o}CO_2+2H_2O\)
\(4FeS_2+11O_2\xrightarrow[]{t^o}2Fe_2O_3+8SO_2\)
\(C_2H_6O+3O_2\xrightarrow[]{t^o}2CO_2+3H_2O\)
C+ O2 -to-> CO2
S+ O2 -to-> SO2
2 Cl2 + 7 O2 -to-> 2 Cl2O7
4P +5 O2 -to-> 2 P2O5
4Al + 3 O2 -to-> 2 Al2O3
4 Na+ O2 -to-> 2 Na2O
Mg + 1/2 O2 -to-> MgO
3 Fe +2 O2 -to-> Fe3O4
4 FeS2 + 11 O2 -to,xt-> 8 SO2 + 2 Fe2O3
H2 + 1/2 O2 -to->H2O
2 Ag + 1/2 O2 -to-> Ag2O
C2H6O + 3 O2 -to-> 2 CO2 + 3 H2O
CH4 + 2 O2 -to-> CO2 + 2 H2O
Cu + 1/2 O2 -to-> CuO

2Ca + O2 ---to→ 2CaO
4Na + O2 ---to→ 2Na2O
S + O2 ---to→ SO2
3Fe + 2O2 ---to→ Fe3O4
C + O2 ---to→ CO2
4Na + O2 ---to→ 2Na2O
2Cu + O2 ---to→ 2CuO
2Zn + O2 ---to→ 2ZnO
2Mg + O2 ---to→ 2MgO
CH4 + 2O2 ---to→ CO2 + 2H2O
2Ba + O2 ---to→ 2BaO

Bài 1: CTHH:
Các hợp chất: K2O, Al2O3, FeO, Fe2O3, Fe3O4, CuO, Cu2O, MgO, Na2O, ZnO, CO, CO2, SO2, SO3, P2O3 , P2O5
\(PTK_{K_2O}=2.39+16=94\left(đ.v.C\right)\)
\(PTK_{Al_2O_3}=2.27+3.16=102\left(đ.v.C\right)\)
\(PTK_{FeO}=56+16=72\left(đ.v.C\right)\\ PTK_{Fe_2O_3}=2.56+3.16=160\left(đ.v.C\right)\\ PTK_{Fe_3O_4}=3.56+4.16=232\left(đ.v.C\right)\)
\(PTK_{CuO}=64+16=80\left(đ.v.C\right)\\ PTK_{Cu_2O}=2.64+16=144\left(đ.v.C\right)\\ PTK_{MgO}=24+16=40\left(đ.v.C\right)\\ PTK_{Na_2O}=2.23+16=62\left(đ.v.C\right)\\ PTK_{ZnO}=65+16=81\left(đ.v.C\right)\\ PTK_{CO}=12+16=28\left(đ.v.C\right)\\ PTK_{CO_2}=12+2.16=44\left(đ.v.C\right)\\ PTK_{SO_2}=32+2.16=64\left(đ.v.C\right)\\ PTK_{SO_3}=32+3.16=80\left(đ.v.C\right)\\ \)
\(PTK_{P_2O_3}=2.31+3.16=110\left(đ.v.C\right)\\ PTK_{P_2O_5}=2.31+5.16=142\left(đ.v.C\right)\)
Bài 2:
PTHH điều chế các oxit trên:
(1) CO2
PTHH: C + O2 -to-> CO2
hoặc Fe2O3 + 3 CO -to-> 2 Fe + 3 CO2
(2) SO2
PTHH: S + O2 -to-> SO2
hoặc Cu +2 H2SO4(đ) -to-> CuSO4 + 2 H2O + SO2
(3) P2O5
PTHH: 4 P + 5 O2 -to-> 2 P2O5
(4) Al2O3
PTHH: 4Al + 3 O2 -to-> 2 Al2O3
(5) Fe3O4
PTHH: 3 Fe +2 O2 -to-> Fe3O4
(6) H2O
PTHH: 2 H2 + O2 -to-> 2 H2O
(7) CuO
PTHH: 2 Cu + O2 -to-> 2 CuO
(8) K2O
PTHH: 4 K + O2 -to-> 2 K2O

Bổ sung
Nhiệt phân thu được khí O2: KMnO4, KClO3.
2KMnO4 \(\underrightarrow{^{to}}\) K2MnO4 + MnO2 + O2
2KClO3 \(\underrightarrow{^{to}}\) 2KCl + 3O2
Tác dụng với nước ở nhiệt độ thường: SO3, P2O5, Ca, BaO
SO3 + H2O \(\rightarrow\) H2SO4
P2O5 + 3H2O \(\rightarrow\) 2H3PO4
Ca + 2H2O\(\rightarrow\) Ca(OH)2 + H2
BaO + H2O \(\rightarrow\) Ba(OH)2
Tác dụng với oxi ở đk thích hợp: Ca, CH4, Fe.
2Ca + O2 \(\underrightarrow{^{to}}\) 2CaO
CH4 + 2O2 \(\underrightarrow{^{to}}\) CO2 + 2H2O
3Fe + 2O2 \(\underrightarrow{^{to}}\) Fe3O4.
Link nek:https://hoc24.vn/hoi-dap/tim-kiem?q=Cho+c%C3%A1c+ch%E1%BA%A5t+:+KMnO4+,+CO2+,+H2O+,+SO3+,+HgO+,+NaNO3+,+Na+,+Ca+,+P2O5+,+BaO+,+Mn2O5+,+CH4+,+SO2+,+Fe+.+H%E1%BB%8Fi+trong+s%E1%BB%91+c%C3%A1c+ch%E1%BA%A5t+tr%C3%AAn+,+c%C3%B3+nh%E1%BB%AFng+ch%E1%BA%A5t+n%C3%A0o+:++1+.+Nhi%E1%BB%87t+ph%C3%A2n+thu+%C4%91%C6%B0%E1%BB%A3c+O2+++2+.+T%C3%A1c+d%E1%BB%A5ng+%C4%91%C6%B0%E1%BB%A3c+v%E1%BB%9Bi+H2O+%E1%BB%9F+nhi%E1%BB%87t+%C4%91%E1%BB%99+th%C6%B0%E1%BB%9Dng+,+t%C3%A1c+d%E1%BB%A5ng+v%E1%BB%9Bi+n%C6%B0%E1%BB%9Bc+v%C3%B4i+trong+,+t%C3%A1c+d%E1%BB%A5ng+v%E1%BB%9Bi+oxi+%E1%BB%9F+nhi%E1%BB%87t+%C4%91%E1%BB%8D+th%C3%ADch+h%E1%BB%A3p&id=229647

a/ 4Al+3O2→Al2O3
2Zn+O2→2ZnO
3Fe+2O2→Fe3O4
2Cu+O2→2CuO
4Na+O2→2Na2O
C+O2→CO2
S+O2→SO2
4P+5O2→2P2O5

a) metan cháy trong oxi tạo thành khí cacbonic và hơi nước
b) CH4+ 2O2 ----> CO2 + 2H2O
c) tỉ lệ số phân tử metan với số phân tử oxi =1/2.
a) metan cháy trong oxi sinh ra khí cacbonic và nước
b)CH4+O2--->CO2+H2O
CH4+2O2--->2H2O+CO2
c)tỉ lệ số phân phân tử metan với số nguyên tử oxi là 1/2
![]()

4Na + O2\(\underrightarrow{t^o}\) 2Na2O
2Mg + O2 \(\underrightarrow{t^o}\) 2MgO
4Al + 3O2 \(\underrightarrow{t^o}\) 2Al2O3
3Fe + 2O2 \(\underrightarrow{t^o}\) Fe3O4
2Cu + O2 \(\underrightarrow{t^o}\) 2CuO
C + O2 \(\underrightarrow{t^o}\) CO2
S + O2 \(\underrightarrow{t^o}\) SO2
4P + 5O2 \(\underrightarrow{t^o}\) 2P2O5
2H2 + O2 \(\underrightarrow{t^o}\) 2H2O
2CO + O2 \(\underrightarrow{t^o}\) 2CO2
4K + O2 \(\underrightarrow{t^o}\) 2K2O
CH4 + 2O2 \(\underrightarrow{t^o}\) CO2 + 2H2O
2C6H6 + 15O2 \(\underrightarrow{t^o}\) 12CO2 + 6H2O
C2H6O + 3O2 \(\underrightarrow{t^o}\) 2CO2 + 3H2O
2C4H10 + 13O2 \(\underrightarrow{t^o}\) 8CO2 + 10H2O
C2H4 + 3O2 \(\underrightarrow{t^o}\) 2CO2 + 2H2O
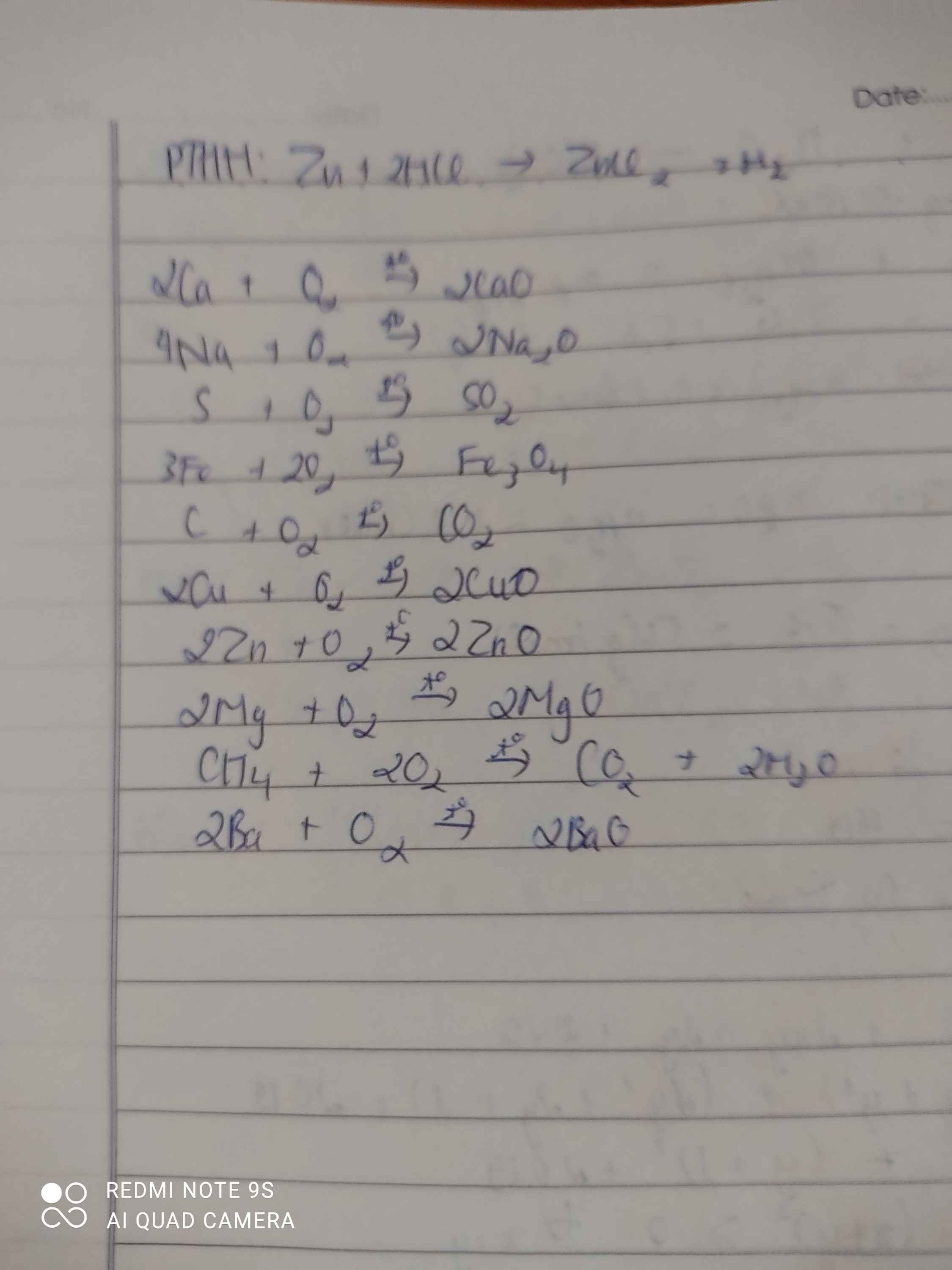
a, \(4Al+3O_2\rightarrow Al_2O_3\)
\(2Zn+O_2\rightarrow2ZnO\)
\(3Fe+2O_2\rightarrow Fe_3O_4\)
\(2Cu+O_2\rightarrow2CuO\)
\(4Na+O_2\rightarrow2Na_2O\)
\(C+O_2\rightarrow CO_2\)
\(S+O_2\rightarrow SO_2\)
\(4P+5O_2\rightarrow2P_2O_5\)
b, \(2CO+O_2\rightarrow2CO_2\)
\(CH_4+2O_2\rightarrow CO_2+2H_2O\)
\(2C_2H_2+5O_2\rightarrow4CO_2+2H_2O\)
\(C_2H_6O+3O_2\rightarrow2CO_2+3H_2O\)