Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, Ta có: \(n_{Fe}=\dfrac{5,6}{56}=0,1\left(mol\right)\)
PT: \(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
Theo PT: \(n_{FeSO_4}=n_{Fe}=0,1\left(mol\right)\Rightarrow m_{FeSO_4}=0,1.152=15,2\left(g\right)\)
b, \(n_{H_2}=n_{Fe}=0,1\left(mol\right)\Rightarrow V_{H_2}=0,1.22,4=2,24\left(l\right)\)
c, Sửa đề: 500 ml → 500 (g)
Theo PT: \(n_{H_2SO_4}=n_{Fe}=0,1\left(mol\right)\Rightarrow C\%_{H_2SO_4}=\dfrac{0,1.98}{500}.100\%=1,96\%\)

nH2=0,15 mol
2Al+3H2SO4=>Al2(SO4)3+3H2
0,1 mol<= 0,15 mol
mAl=0,1.27=2,7g
nAl2(SO4)3=0,05 mol
=>mAl2(SO4)3=342.0,05=17,1g
nH2SO4=0,15 mol=>mH2SO4=14,7
mdd H2SO4=14,7/10%=147g
mdd sau pứ=2,7+147-0,15.2=149,4g
C%dd Al2(SO4)3=17,1/149,4.100%=11,45%

a, \(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
\(n_{H_2SO_4}=0,2.1,35=0,27\left(mol\right)\)
PT: \(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
Xét tỉ lệ: \(\dfrac{0,2}{2}>\dfrac{0,27}{3}\), ta được Al dư.
Theo PT: \(n_{H_2}=n_{H_2SO_4}=0,27\left(mol\right)\Rightarrow V_{H_2}=0,27.22,4=6,048\left(l\right)\)
b, \(n_{Al\left(pư\right)}=\dfrac{2}{3}n_{H_2SO_4}=0,18\left(mol\right)\)
\(\Rightarrow m_{Al\left(pư\right)}=0,18.27=4,86\left(g\right)\)
c, \(n_{Al_2\left(SO_4\right)_3}=\dfrac{1}{3}n_{H_2SO_4}=0,09\left(mol\right)\)
\(\Rightarrow C_{M_{Al_2\left(SO_4\right)_3}}=\dfrac{0,09}{0,2}=0,45\left(M\right)\)

n hh khí = 0.5 mol
nCO: x mol
nCO2: y mol
=> x + y = 0.5
28x + 44y = 17.2 g
=> x = 0.3 mol
y = 0.2 mol
Khối lượng oxi tham gia pứ oxh khử oxit KL: 0.2 * 16 = 3.2g => m KL = 11.6 - 3.2 = 8.4g
TH: KL hóa trị I => nKL = 2*nH2 = 0.3 mol => KL: 28!!
KL hóa trị III => nKL = 2/3 *nH2 = 0.1 mol => KL: 84!!
KL hóa trị II => nKL = nH2 = 0.15 mol => KL: 56 => Fe.
nFe / Oxit = 0.15 mol
nO/Oxit = 0.2 mol
=> nFe/nO = 3/4 => Fe3O4
Fe3O4 + 4CO = 3Fe + 4CO2
Fe + H2SO4 = FeSO4 + H2
0.15.....0.15.......0.15.....0.15
=> mH2SO4 pứ = 14.7 g => mdd = 147 g
m dd sau khi cho KL vào = m KL + m dd - mH2 thoát ra = 0.15 * 56 + 147 - 0.15*2 = 155.1g
=> C% FeSO4 = 14.7%

a, \(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
b, \(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
Theo PT: \(n_{H_2SO_4}=n_{Fe}=0,2\left(mol\right)\Rightarrow m_{H_2SO_4}=0,2.98=19,6\left(g\right)\)
c, \(C\%_{H_2SO_4}=\dfrac{19,6}{50}.100\%=39,2\%\)
d, Theo PT: \(n_{H_2}=n_{Fe}=0,2\left(mol\right)\Rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)

PTHH: 2Na+2H2O=>2 NaOH+H2
nH2SO4=0,2mol
PTHH: 2NaOH+H2SO4=> Na2SO4+2H2O
0,4mol<-0,2mol
=> n NaOH=0,4mol
mà nNaOH=nNa=0,4mol
=> m Na =0,4.23=9,2g
nH2=1/2nNaOH=1/2.0,2=0,1mol
=> V H2=0,1.22,4=2,24ml

PTHH: \(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\uparrow\)
Ta có: \(\left\{{}\begin{matrix}n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\\n_{H_2SO_4}=\dfrac{200\cdot29,4\%}{98}=0,6\left(mol\right)\end{matrix}\right.\)
Xét tỉ lệ: \(\dfrac{0,2}{2}< \dfrac{0,6}{3}\) \(\Rightarrow\) Axit còn dư, Nhôm p/ứ hết
\(\Rightarrow\left\{{}\begin{matrix}n_{Al_2\left(SO_4\right)_3}=0,1\left(mol\right)\\n_{H_2}=0,3\left(mol\right)\\n_{H_2SO_4\left(dư\right)}=0,6-0,3=0,3\left(mol\right)\end{matrix}\right.\)\(\Rightarrow\left\{{}\begin{matrix}m_{Al_2\left(SO_4\right)_3}=0,1\cdot342=34,2\left(g\right)\\m_{H_2SO_4\left(dư\right)}=0,3\cdot98=29,4\left(g\right)\\m_{H_2}=0,3\cdot2=0,6\left(g\right)\\V_{H_2}=0,3\cdot22,4=6,72 \left(l\right)\end{matrix}\right.\)
Mặt khác: \(m_{dd\left(saup/ứ\right)}=m_{Al}+m_{ddH_2SO_4}-m_{H_2}=204,8\left(g\right)\)
\(\Rightarrow\left\{{}\begin{matrix}C\%_{Al_2\left(SO_4\right)_3}=\dfrac{34,2}{204,8}\cdot100\%\approx16,7\%\\C\%_{H_2SO_4\left(dư\right)}=\dfrac{29,4}{204,8}\cdot100\%\approx14,36\%\end{matrix}\right.\)

\(n_{Al}=\frac{10,8}{27}=0,4\left(mol\right)\)
\(2Al+6HCl->2AlCl_3+3H_2\) (1)
theo (1) \(n_{H_2}=\frac{3}{2}n_{Al}=0,6\left(mol\right)\)
=> \(V_{H_2}=0,6.22,4=13,44\left(l\right)\)
Cho 10,08 g nhom tac dung vua du voi dung dich axit HCl.2M
a) viet phuong trinh phan ung va tinh the tich H2(dktc)
b) tinh the tich dung dich axit HCl.2M da dung
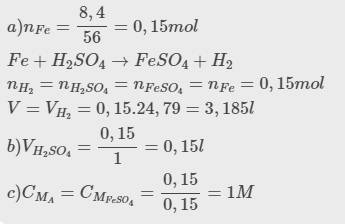
\(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
PT: \(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
Theo PT: \(n_{H_2}=\dfrac{3}{2}n_{Al}=0,3\left(mol\right)\)
\(\Rightarrow V_{H_2}=0,3.24,79=7,437\left(l\right)\)
\(n_{Al}=\dfrac{5,4}{27}=0,2mol\\ 2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\\ n_{H_2}=1,5.0,2=0,3mol\\ V_{H_2}=0,3.24,79=7,437l\)