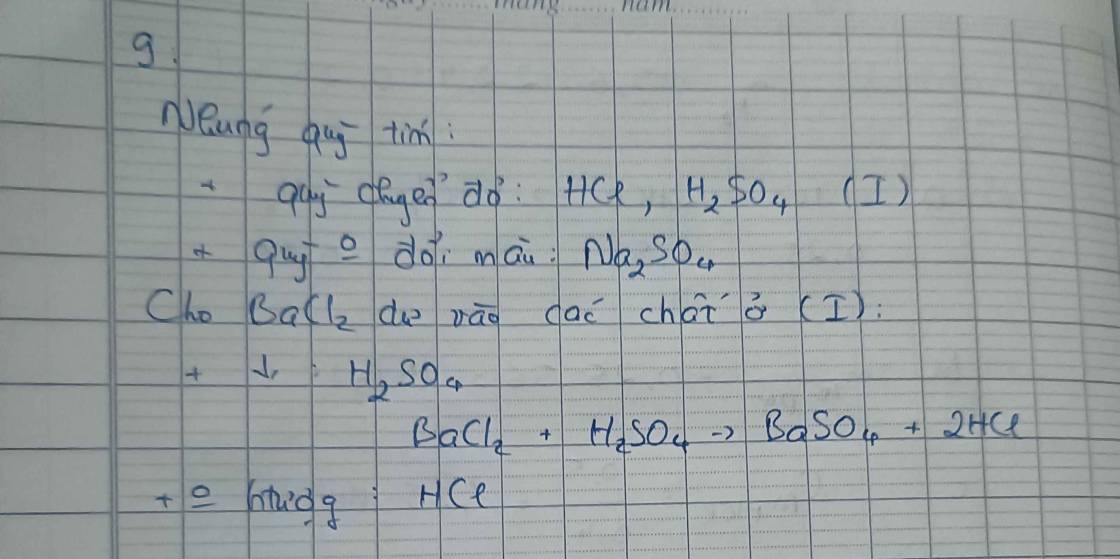Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

thầy mik cho thiếu, mik vừa sửa lại nếu b lm đc mong b giúp mik ;.;

\(n_{HCl}=6\cdot0,05=0,3\left(mol\right)\\ a,PTHH:2Al+6HCl\rightarrow2AlCl_3+3H_2\)
Hiện tượng: Al tan dần, có bọt khí không màu xuất hiện
\(b,\left\{{}\begin{matrix}n_{Al}=\dfrac{1}{3}n_{HCl}=0,1\left(mol\right)\\n_{H_2}=\dfrac{1}{2}n_{HCl}=0,15\left(mol\right)\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}m_{Al}=0,1\cdot27=2,7\left(g\right)\\V_{H_2\left(đkc\right)}=0,15\cdot24,79=3,7185\left(l\right)\end{matrix}\right.\)

a) 2Al+6HCl→→2AlCl3+3H2
b)
nAl=10,8\27=0,4(mol)
nAlCl3=nAl=0,4(mol)
mAlCl3=0,4.133,5=53,4(g)
c)
nH2=3\2nAl=0,6(mol)
VH2=22,4.0,6=13,44(l)
d) n HCl=0,4.6\2=1,2 mol
=>Cm HCl=1,2\0,1=12M
\(n_{Al}=\dfrac{10,8}{27}=0,4\left(mol\right)\)
a) Pt : \(2Al+6HCl\rightarrow2AlCl_3+3H_2|\)
2 6 2 3
0,4 1,2 0,4 0,6
b) \(n_{H2}=\dfrac{0,4.3}{2}=0,6\left(mol\right)\)
\(V_{H2\left(dktc\right)}=0,6.24,79=14,874\left(l\right)\)
c) \(n_{AlCl3}=\dfrac{0,6.2}{3}=0,4\left(mol\right)\)
⇒ \(m_{AlCl3}=0,4.133,5=53,4\left(g\right)\)
d) \(n_{HCl}=\dfrac{0,4.6}{2}=1,2\left(mol\right)\)
100ml = 0,1l
\(C_{M_{ddHCl}}=\dfrac{1,2}{0,1}=12\left(M\right)\)
Chúc bạn học tốt

\(a,n_{Mg}=\dfrac{4,8}{24}=0,2\left(mol\right)\\ Mg+2HCl\rightarrow MgCl_2+H_2\\ b,n_{MgCl_2}=n_{H_2}=n_{Mg}=0,2\left(mol\right)\\ b,V_{H_2\left(25\text{đ}\text{ộ}C,1bar\right)}=0,2.24,79=4,958\left(l\right)\\ c,n_{HCl}=2.0,2=0,4\left(mol\right)\\ m_{\text{dd}HCl}=\dfrac{0,4.36,5.100}{10}=146\left(g\right)\\ m_{\text{dd}A}=m_{Mg}+m_{\text{dd}HCl}-m_{H_2}=4,8+146-0,2.2=150,4\left(g\right)\\ d,C\%_{\text{dd}MgCl_2}=\dfrac{95.0,2}{150,4}.100\approx12,633\%\)

\(a,PTHH:Fe+2HCl\rightarrow FeCl_2+H_2\\ n_{Fe}=\dfrac{22,4}{56}=0,4\left(mol\right)\\ b,n_{FeCl_2}=n_{H_2}=n_{Fe}=0,4\left(mol\right)\\ V_{H_2\left(đktc\right)}=0,4.22,4=8,96\left(l\right)\\ c,m_{FeCl_2}=127.0,4=50,8\left(g\right)\)

\(n_{Mg}=\dfrac{4,8}{24}=0,2\left(mol\right)\)
PTHH: Mg + 2HCl → MgCl2 + H2
Mol: 0,2 0,4 0,2
\(V_{H_2}=0,2.24,79=4,958\left(l\right)\)
\(m_{HCl}=0,4.36,5=14,6\left(g\right)\Rightarrow m_{ddHCl}=\dfrac{14,6.100\%}{20\%}=73\left(g\right)\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\\ n_{Mg}=\dfrac{4,8}{24}=0,2\left(mol\right)\\ n_{H_2}=n_{Mg}=0,2\left(mol\right)\\ V_{H_2\left(25^oC,1bar\right)}=0,2.22,4=4,48\left(l\right)\\ n_{HCl}=0,2.2=0,4\left(mol\right)\\ m_{HCl}=0,4.36,5=14,6\left(g\right)\)

Ta có: \(n_{CuO}=\dfrac{8}{80}=0,1\left(mol\right)\)
PT: \(CuO+2HCl\rightarrow CuCl_2+H_2O\)
a, Theo PT: \(n_{CuCl_2}=n_{CuO}=0,1\left(mol\right)\Rightarrow m_{CuCl_2}=0,1.135=13,5\left(g\right)\)
b, \(n_{HCl}=2n_{CuO}=0,2\left(mol\right)\Rightarrow C_{M_{HCl}}=\dfrac{0,2}{0,2}=1\left(M\right)\)